Transcranial magnetic stimulation (TMS) is FDA-cleared for treating Major Depressive Disorder, especially when medications haven’t worked well. It’s also cleared for Obsessive-Compulsive Disorder, migraine relief, and helping people quit smoking. These specific uses show where TMS has proven benefits, backed by research and clinical trials.
If you’re thinking about TMS, knowing these approved conditions helps set realistic expectations. There are other possible uses being studied, but they’re not yet official. Keep reading to learn more about how TMS works, who might qualify, and whether it fits your treatment plan.
Key Takeaway
- TMS is FDA-cleared for treatment-resistant depression, OCD, migraines, and smoking cessation.
- Its use for other conditions, like anxiety or PTSD, is often considered off-label.
- Patient eligibility is strict, often requiring prior treatment failure and the absence of specific health risks.
The Foundation of TMS Therapy
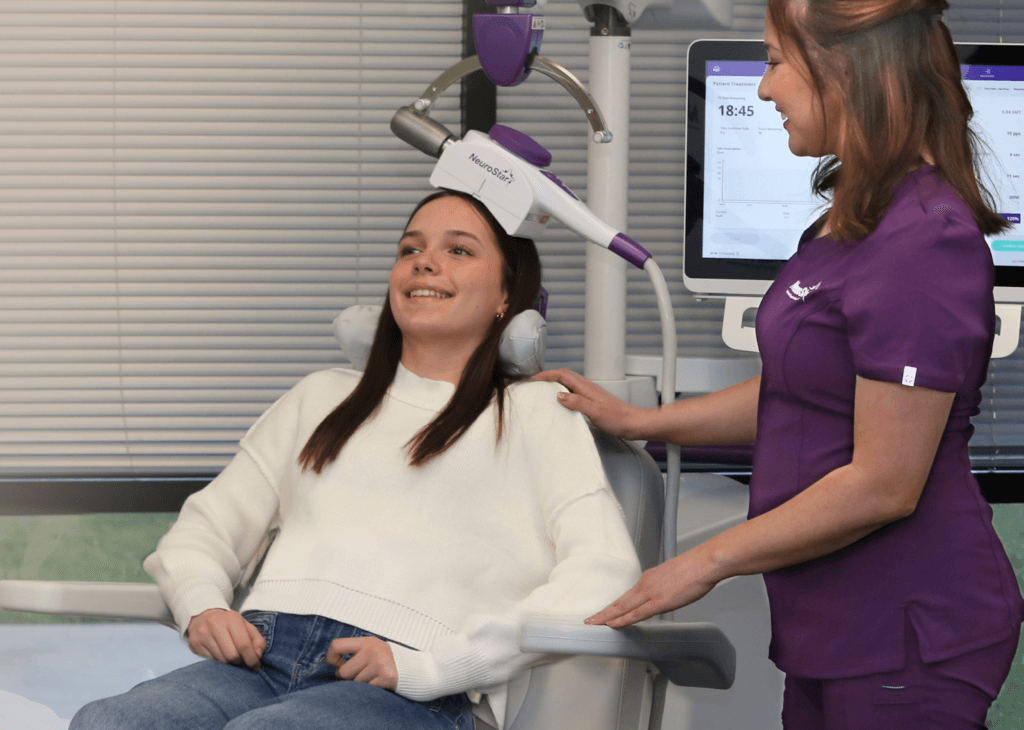
Transcranial Magnetic Stimulation (TMS) is a new way to treat some brain-related health problems, with systems like NeuroStar Advanced TMS Therapy offering a precise, non-invasive approach. It doesn’t need surgery or implants. Instead, it uses magnetic pulses to gently stimulate specific parts of the brain. These parts are chosen because they tend to be less active in certain mental health conditions.
Unlike medicines that affect the whole body, TMS works directly on the brain areas that control mood and other important functions. This focused approach is why the FDA has cleared TMS for some conditions when regular treatments don’t work well enough.
TMS is especially useful for people with Major Depressive Disorder who haven’t improved with antidepressant medicines or can’t handle their side effects. It offers another option when other treatments have failed. Most people tolerate TMS well, with only mild and temporary side effects.
In short, TMS is a precise, non-invasive treatment that helps adjust brain activity linked to certain disorders. It gives doctors and patients another tool to fight conditions that don’t respond to usual therapies.
FDA-Cleared Condition: Major Depressive Disorder

For people with Major Depressive Disorder (MDD), Transcranial Magnetic Stimulation (TMS) is usually recommended when antidepressant medications have not worked well enough. This often means trying at least two different antidepressants without enough improvement. When this happens, doctors consider the depression treatment-resistant.
TMS targets a part of the brain called the left dorsolateral prefrontal cortex. This area helps control mood and thinking. The treatment uses magnetic pulses to stimulate this part of the brain, aiming to improve its activity and help ease depression symptoms.
Studies have shown that TMS can help many patients who didn’t get better with medication, supported by a multisite randomized controlled trial showing significantly greater improvement than sham stimulation [1]. Some people see their symptoms improve a lot, and some even reach full remission.
The treatment usually happens every day for a few weeks. It is safe and most people have only mild side effects. For those struggling with depression that doesn’t respond to medicine, TMS offers a new way to find relief and improve quality of life.
- Primary Indication: Treatment-resistant Major Depressive Disorder.
- Typical Patient Profile: Adults who have not responded to at least two antidepressant trials.
- Treatment Goal: Symptom relief and achieving remission from depressive episodes.
The protocol involves daily sessions over several weeks. Patients remain awake and alert during the procedure, which lasts about 19 minutes. There is no recovery time needed, allowing individuals to resume their daily activities immediately afterward. This makes it a practical option for those maintaining work or family schedules.
FDA-Cleared Condition: Obsessive-Compulsive Disorder
The FDA-cleared Transcranial Magnetic Stimulation (TMS) for Obsessive-Compulsive Disorder (OCD) after careful study, much like its approval for depression. This treatment is meant for adults who have not seen enough improvement from standard therapies. These usually include medications called serotonin reuptake inhibitors (SRIs) and cognitive-behavioral therapy (CBT), which are the first-line treatments for OCD.
Unlike depression, TMS for OCD targets different brain areas. The focus is on the medial prefrontal cortex and the anterior cingulate cortex. These regions are linked to the intrusive thoughts and repetitive behaviors that define OCD. By stimulating these specific brain circuits, TMS aims to reduce the intensity of these symptoms.
Clinical trials have found that TMS can significantly lower the severity of OCD symptoms, a benefit reflected in dedicated programs such as TMS therapy for OCD. It is important to understand that TMS is not a cure, but it can be a valuable part of managing the disorder, especially when other treatments have not worked well enough.
With a meta-analysis of 20 randomized sham-controlled trials showing a small-to-moderate reduction in symptom severity [2], a benefit reflected in dedicated programs such as TMS therapy for OCD.
The treatment schedule is like that used for depression, with many sessions each week over several weeks.
TMS offers a targeted, non-invasive option for adults struggling with OCD who need more help beyond standard therapies.
Other FDA-Cleared Applications

Transcranial Magnetic Stimulation (TMS) has received FDA clearance for several conditions beyond depression and Obsessive-Compulsive Disorder (OCD). One important use is for migraine headaches. A specialized TMS device is cleared for treating migraines with aura as they occur and for preventing future attacks.
The treatment works by calming overactive nerve cells in the brain’s cortex. This helps stop a migraine from fully developing. For patients who prefer to avoid medications or who cannot tolerate them, TMS offers a non-drug option to manage migraines.
TMS is also cleared to support smoking cessation. In this case, the treatment targets brain areas involved in addiction and craving. By stimulating these regions, TMS can help reduce the urge to smoke and ease withdrawal symptoms. It is important to note that TMS for smoking cessation is meant to be part of a larger treatment plan, which includes counseling and behavioral support. It is not intended to be used alone.
In both migraine and smoking cessation, TMS provides a new approach that works directly on brain activity. This expands the range of tools doctors can use to help patients with these challenging conditions.
Exploring Off-Label and Investigational Uses
In medical practice, Transcranial Magnetic Stimulation (TMS) is sometimes used “off-label,” a topic explored in this advanced therapy that outlines how doctors may recommend it for conditions not yet FDA-cleared, based on emerging research and clinical judgment.
One common off-label use is for anxiety disorders, like generalized anxiety and panic disorder. These often happen together with depression, and TMS may help when other treatments don’t work well enough.
TMS is also being studied for Posttraumatic Stress Disorder (PTSD). Early studies suggest it might help by affecting brain areas that control fear and emotions, which are often out of balance in PTSD. More research is needed to know for sure.
For bipolar depression, TMS has a special FDA status called “breakthrough device.” This speeds up the process to develop and review TMS for this condition because it’s hard to treat. But full FDA clearance for bipolar depression is still in progress.
Using TMS off-label shows how doctors try to use new evidence to help patients, even before official approvals catch up. It’s done carefully, with attention to safety and effectiveness.
- Common Off-Label Uses: Anxiety disorders, PTSD.
- Investigational Areas: Bipolar depression, chronic pain, stroke rehabilitation.
- Important Note: Off-label use requires a detailed discussion with your doctor about the evidence, potential benefits, and risks.
These applications are supported by a growing body of research. Yet, insurance coverage for off-label treatments can be more variable and often requires a strong justification from the treating physician. It underscores the importance of a thorough consultation with a TMS provider.
Determining Your Eligibility for TMS
Not everyone is eligible for Transcranial Magnetic Stimulation (TMS). Whether a person can receive this treatment depends on their diagnosis and individual health factors. For conditions like depression or Obsessive-Compulsive Disorder (OCD), the main need is usually a history showing that standard treatmentslike medications or therapyhave not worked well enough.
Before starting TMS, a qualified healthcare provider will perform a thorough evaluation. This includes confirming the diagnosis and reviewing past treatments to ensure TMS is appropriate.
Safety is a key concern when deciding if someone can have TMS. Patients with metal implants in or near the head such as aneurysm clips, cochlear implants, or deep brain stimulators are not candidates. The magnetic pulses used in TMS can interfere with these devices or cause harm. Additionally, a personal or family history of seizures is an important factor. TMS carries a small risk of triggering a seizure, so people with this history need careful evaluation and may be excluded.
These safety rules are in place to protect patients from potential harm. The goal is to offer TMS only when it is both safe and likely to be effective, ensuring the best possible outcome.
FAQ
How does TMS work in the brain, and why is it used for treatment resistant depression?
TMS uses magnetic stimulation for depression by sending gentle pulses to the left dorsolateral prefrontal cortex. This area helps regulate mood. Researchers study repetitive transcranial magnetic stimulation in TMS clinical trials to understand how depression neurocircuitry changes. Many people with treatment resistant depression try this non-invasive depression treatment when medication fails, hoping for steady depression symptom relief and better daily functioning.
What should I know about TMS side effects and the overall depression treatment side effect profile?
TMS side effects are usually mild, like small headaches or scalp tingling. This non-pharmacological depression treatment avoids many medication issues. Because brain magnetic pulse therapy targets the left prefrontal cortex stimulation area, it doesn’t affect the whole body. Long-term TMS therapy guidelines track safety, neuropsychiatric treatment options, and the neuromodulation safety profile across many TMS efficacy studies.
How do rTMS session protocols support depression symptom improvement measures?
High-frequency rTMS directs repetitive magnetic stimulation benefits toward mood control areas. TMS session protocols often use left DLPFC in depression because this region supports emotional balance. Doctors watch clinical response to TMS, depression symptom clusters, and depression treatment outcomes over time. Many people see steady depression symptom improvement measures, especially in treatment resistant mood disorders and major depressive disorder treatment.
Can TMS help people who have resistant depression management needs or want depression medication alternatives?
Many people with treatment for refractory depression explore TMS for anxiety and depression as part of resistant depression management. This brain stimulation therapy supports neuroplasticity and TMS, helping mood disorder neuromodulation without drugs. Some people choose it as adjunctive depression treatment or as part of non-medication depression alternatives. It can complement depression treatment innovations focused on personalized care and clinical depression biomarkers.
What long-term factors matter most in TMS maintenance therapy and relapse prevention?
TMS maintenance therapy focuses on depression relapse prevention, treatment adherence in depression, and quality of life post-TMS. Studies track TMS remission rates, depression treatment personalization, and clinical depression relapse patterns. Researchers also study cognitive effects of TMS, TMS and cognitive function, and TMS long-term effects. These help guide TMS therapy clinical guidelines, TMS insurance coverage decisions, and neurophysiology of depression planning.
What Conditions Is TMS Approved For and Why It Matters
TMS is FDA-cleared for Major Depressive Disorder, OCD, migraine, and smoking cessation. For other conditions, the decision depends on clinical judgment and emerging research. Consulting a psychiatrist or neurologist experienced in neuromodulation is essential to test your history and symptoms. If you’ve faced treatment-resistant depression or OCD, discussing TMS could be a vital step. Reach out to a certified provider to start the conversation: TMS of the Carolinas.
References
- https://ajp.psychiatryonline.org/doi/full/10.1176/appi.ajp.2007.06111860
- https://www.sciencedirect.com/science/article/pii/S2213158222002563
